Solubility of Salts
Which is Which?
Use the table below to help you identify which salts are soluble and which are not.
| Soluble | Insoluble |
|---|---|
| All Sodium, Potassium and ammonium | |
| Chlorides | Except silver and lead, chlorides |
| All Ethanoates | |
| Sulphate | Except calcium, barium and lead sulphates |
| All Nitrates | |
| Sodium, potassium and ammonium carbonates and hydroxides | All other carbonates and hydroxides are insoluble |
Preparation of Soluble Salts
To prepare soluble salts you must form crystals of them.
Preparing Insoluble Salts
A precipitate is the name given to a solid salt that forms when an insoluble salt is produced from a reaction. For example, with barium chloride and magnesium sulphate...
Barium Chloride + Magnesium Sulphate  Barium Sulphate + Magnesium Chloride
Barium Sulphate + Magnesium Chloride
BaCl2 + MgSO4  BaSO4 + MgCl2
BaSO4 + MgCl2
You will see a white precipitate of barium sulphate ...
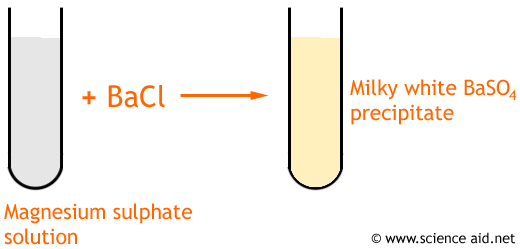
To get a pure, dry sample of Barium Sulphate it must be...
filtered,
washed with distilled water,
and then left to allow the excess water to evaporate.
